Niamh was delighted to collaborate with Dr Terence Capellini’s group in Harvard and contribute to their work on how changes in the ilium contributed to the evolution of bipedalism.
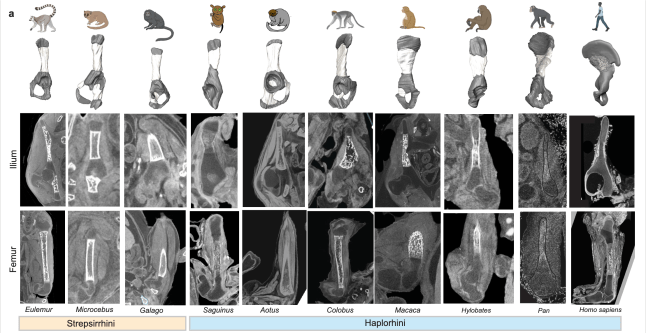
The upper part of the human pelvis, known as the ilium, underwent two major structural innovations during evolution that enabled humans to walk on two legs, a Nature paper reveals. The study lays the developmental and genetic groundwork for the human-defining trait of bipedalism. The ilium is the big, flared part of the pelvis that anchors the powerful gluteus maximus muscles that humans use to stay upright. Differences in the illum between humans and other hominins are a defining evolutionary feature. However, the developmental processes that led to the unique human ilium shape remain unknown.
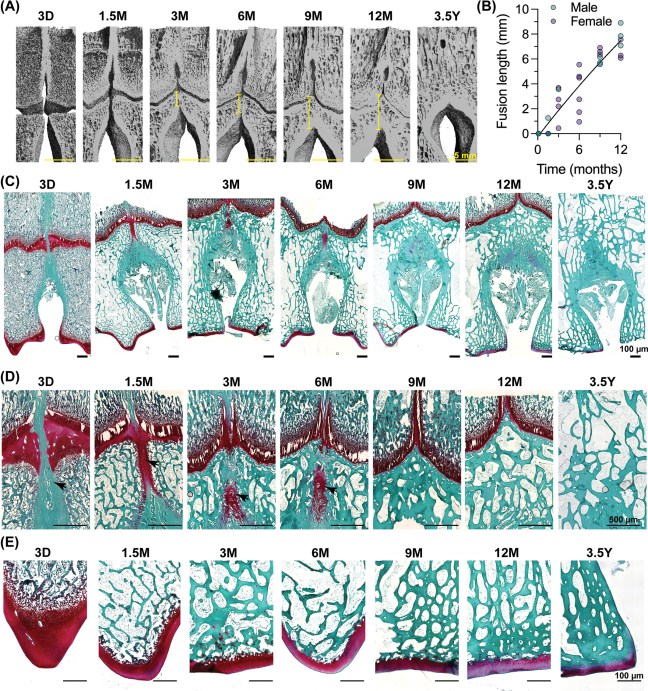
The team, led by Dr Terence Capellini, Harvard University, used histological, anatomical, and functional genomic approaches to reveal how the structure acquired its unique shape. One key change involved the direction of cartilage growth. A shift in the orientation of the cartilage growth plate (physis) enabled the ilium to sit perpendicular to the direction seen in ilia from other animals. A second key change involved the process of bone formation. The authors identify timing and spatial differences in the way that bone cells are laid down over the cartilage in the human ilium, compared with non-human primate ilia and human long bones. Both innovations are interconnected at tissue and molecular levels. Hundreds of regulatory sequences were identified that are active during the development of the human ilium. These show evidence of human evolutionary change, suggesting that key complex, interacting sequences were selected for through time to endow the human pelvis with its unique shape.
Niamh’s involvement in the study centred on a dataset of 3D postmortem imaging data from human embryos and fetuses that she had assemebled together with Prof Owen Arthurs, a paediatric radiologist from Great Ormond Street Hospital in London and UCLH, to characterise hip development in 3D. When Niamh met Terry at a conference, he was excited to hear that Owen and I had a dataset that his team could use to characterise how the shape and ossification of the ilium changes over early human development. His team linked this with their extensive data and research on pelvic development in the human embryo and in other animals.
Read the paper here.